NEXVIAZYME: An M6P-Enriched ERT1,2
Engineered with ~15x more M6P than alglucosidase alfa

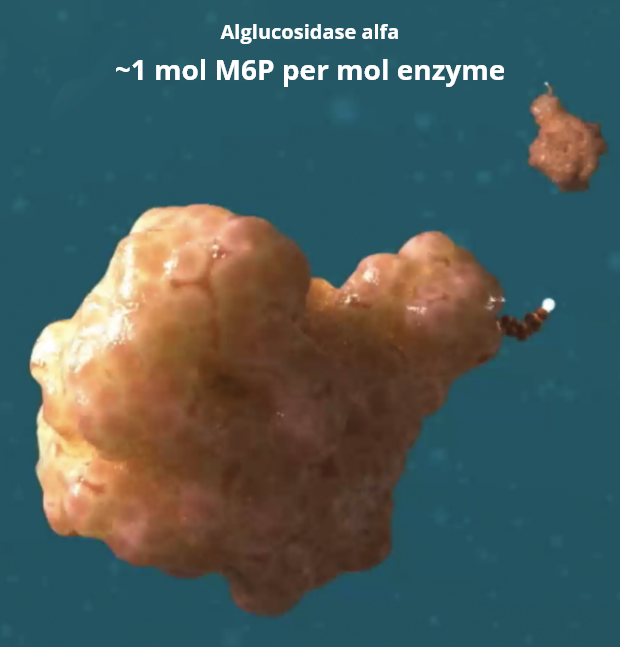

What is M6P?
Mannose-6-phosphate, or M6P, is a residue that binds to muscle cell receptors (called CI-MPRs), mediating the uptake of ERT into muscle cells.3-5
NEXVIAZYME enters the cell via the M6P pathway1,3,5,6
The M6P pathway is a common route for ERTs, and NEXVIAZYME depends on M6P to drive muscle cell uptake.
Once NEXVIAZYME enters the body by intravenous (IV) infusion1:
|
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS Hypersensitivity Reactions including Anaphylaxis Patients treated with NEXVIAZYME have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Appropriate medical support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, NEXVIAZYME should be discontinued immediately and appropriate medical treatment should be initiated. In patients with severe hypersensitivity reaction, a desensitization procedure to NEXVIAZYME may be considered. Infusion-Associated Reactions (IARs) Patients treated with NEXVIAZYME have experienced severe IARs. If severe IARs occur, consider immediate discontinuation of NEXVIAZYME, initiation of appropriate medical treatment, and the benefits and risks of readministering NEXVIAZYME following severe IARs. Patients with an acute underlying illness at the time of NEXVIAZYME infusion may be at greater risk for IARs. Patients with advanced Pompe disease may have compromised cardiac and respiratory function, which may predispose them to a higher risk of severe complications from IARs. Risk of Acute Cardiorespiratory Failure in Susceptible Patients Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function for whom fluid restriction is indicated may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. More frequent monitoring of vitals should be performed during NEXVIAZYME infusion. |
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: See Boxed WARNING. Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. The risks and benefits of readministering NEXVIAZYME following severe hypersensitivity reaction (including anaphylaxis) should be considered. If a mild or moderate hypersensitivity reaction occurs, the infusion rate may be slowed or temporarily stopped.
Infusion-Associated Reactions: See Boxed WARNING. IARs may still occur in patients after receiving pretreatment. If mild or moderate IARs occur regardless of pretreatment, decreasing the infusion rate or temporarily stopping the infusion may ameliorate the symptoms.
Risk of Acute Cardiorespiratory Failure in Susceptible Patients: See Boxed WARNING.
ADVERSE REACTIONS
The most common adverse reactions (>5%) were headache, fatigue, diarrhea, nausea, arthralgia, dizziness, myalgia, pruritus, vomiting, dyspnea, erythema, paresthesia and urticaria.
INDICATION
NEXVIAZYME (avalglucosidase alfa-ngpt) is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease [lysosomal acid alpha-glucosidase (GAA) deficiency].
Please see full Prescribing Information for complete details, including Boxed WARNING.
References: 1. NEXVIAZYME (avalglucosidase alfa-ngpt) [prescribing information]. Genzyme Corporation, Cambridge, MA. 2. Zhu Y, Jiang JL, Gumlaw NK, et al. Mol Ther. 2009;17(6):954-963. 3. Pena LDM, Barohn RJ, Byrne BJ, et al. Neuromuscul Disord. 2019;29(3):167-186. 4. Reuser AJ, Kroos MA, Ponne NJ, et al. Exp Cell Res. 1984;155(1):178-189. 5. Duncan JR, Kornfeld S. J Cell Biol. 1988;106(3):617-628. 6. Baldwin AC, Naatz A, Bohnsack RN, et al. Mol Cell Biol. 2018;38(8):e00680-17.
|
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS Hypersensitivity Reactions including Anaphylaxis Patients treated with NEXVIAZYME have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Appropriate medical support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, NEXVIAZYME should be discontinued immediately and appropriate medical treatment should be initiated. In patients with severe hypersensitivity reaction, a desensitization procedure to NEXVIAZYME may be considered. Infusion-Associated Reactions (IARs) Patients treated with NEXVIAZYME have experienced severe IARs. If severe IARs occur, consider immediate discontinuation of NEXVIAZYME, initiation of appropriate medical treatment, and the benefits and risks of readministering NEXVIAZYME following severe IARs. Patients with an acute underlying illness at the time of NEXVIAZYME infusion may be at greater risk for IARs. Patients with advanced Pompe disease may have compromised cardiac and respiratory function, which may predispose them to a higher risk of severe complications from IARs. Risk of Acute Cardiorespiratory Failure in Susceptible Patients Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function for whom fluid restriction is indicated may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. More frequent monitoring of vitals should be performed during NEXVIAZYME infusion. |
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: See Boxed WARNING. Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. The risks and benefits of readministering NEXVIAZYME following severe hypersensitivity reaction (including anaphylaxis) should be considered. If a mild or moderate hypersensitivity reaction occurs, the infusion rate may be slowed or temporarily stopped.
Infusion-Associated Reactions: See Boxed WARNING. IARs may still occur in patients after receiving pretreatment. If mild or moderate IARs occur regardless of pretreatment, decreasing the infusion rate or temporarily stopping the infusion may ameliorate the symptoms.
Risk of Acute Cardiorespiratory Failure in Susceptible Patients: See Boxed WARNING.
ADVERSE REACTIONS
The most common adverse reactions (>5%) were headache, fatigue, diarrhea, nausea, arthralgia, dizziness, myalgia, pruritus, vomiting, dyspnea, erythema, paresthesia and urticaria.
INDICATION
NEXVIAZYME (avalglucosidase alfa-ngpt) is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease [lysosomal acid alpha-glucosidase (GAA) deficiency].
Please see full Prescribing Information for complete details, including Boxed WARNING.
References: 1. NEXVIAZYME (avalglucosidase alfa-ngpt) [prescribing information]. Genzyme Corporation, Cambridge, MA. 2. Zhu Y, Jiang JL, Gumlaw NK, et al. Mol Ther. 2009;17(6):954-963. 3. Pena LDM, Barohn RJ, Byrne BJ, et al. Neuromuscul Disord. 2019;29(3):167-186. 4. Reuser AJ, Kroos MA, Ponne NJ, et al. Exp Cell Res. 1984;155(1):178-189. 5. Duncan JR, Kornfeld S. J Cell Biol. 1988;106(3):617-628. 6. Baldwin AC, Naatz A, Bohnsack RN, et al. Mol Cell Biol. 2018;38(8):e00680-17.




